Recalled Eye Drops Injuries
There have been reports of serious and grave injuries sustained from recalled eye drops. On February 1, 2023, the Centers for Disease Control and Prevention (CDC) issued a health advisory concerning over-the-counter artificial tear and eye lubricating products distributed by Aru Pharma, EzriCare and Delsam Pharma from Global Pharma Healthcare. The CDC and FDA recommend patients and providers who use the artificial tear products EzriCare and Delsam Pharma stop using them immediately, and Global Pharma Healthcare has issued a recall of the artificial tears products and an artificial eye ointment manufactured by Delsam.
The number of people who have suffered adverse reactions and/or death from defective eye drops continues to increase across the country. If you or a loved one had an adverse reaction to using Global Pharma Healthcare eye products, or if a loved one who used these products has passed away from a pseudomonas aeruginosa-related bacterial infection, please contact PCVA to discuss your options.
What led to the eye drops recall by Global Pharma Healthcare?
From May 2022 to January 2023, the CDC received reports from state and local health departments in California, Colorado, Connecticut, Florida, New Jersey, New Mexico, New York, Nevada, Texas, Utah, Washington and Wisconsin involving consumers’ adverse reactions to EzriCare and Delsam artificial tears. As of April 2023, the number of products recalled, states involved and consumers affected has and will continue to grow. Most recently, the recall was expanded to include other products, including Delsam artificial eye ointment.
What risks do the defective eye drops pose?
Global Pharma Healthcare’s eye drops were found to be contaminated with pseudomonas aeruginosa bacteria, a drug-resistant bacteria that spreads easily, especially in healthcare environments. It can cause severe infections throughout the body (beyond the eyes) and can be especially devastating to those who are immunocompromised.
What is the danger of using the recalled eyedrops?
According to the CDC, the bacteria can cause severe infections throughout the body, including the blood, lungs, stomach and urinary tract. Consumers who used the contaminated products reported eye infections, blurred vision, loss of vision and other issues. Several people have died as well after the infection spread beyond their eyes.
The CDC notes symptoms may include: yellow, green or clear discharge from the eye; eye pain or discomfort; redness of the eye or eyelid; feeling like something is in the eye; increased light sensitivity; and blurry vision.
How do I know if I am using recalled products?
The recalled eye drops and ointment are packaged like the photos below. If you are using them, please stop immediately and call your healthcare provider or eye doctor.
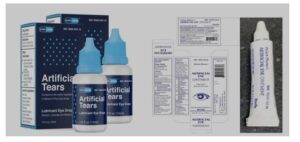
For additional current information on the recalled products, you are encouraged to view the FDA recall page, the Global Pharma Healthcare website for the eye drops and the FDA recall page for the recalled eye ointment.
I purchased recalled eyedrops. What should I do?
If you purchased the recalled eyedrops, the most important thing to do is stop using them immediately and contact your healthcare provider or eye doctor. After that, consult a qualified personal injury attorney to discuss your case to determine next steps.
How can PCVA help if you have been injured by Global Pharma Healthcare artificial tears?
PCVA’s experienced attorneys can help you understand your options for pursuing compensation and damages from those responsible for the injuries you suffered after exposure to these dangerous eyedrops. If you would like to speak with a PCVA lawyer, complete our online form or call us at (253) 948-3199 or (206) 536-2850.
How much does PCVA charge?
Our work is done on a contingency basis. This means that you do not pay us on an hourly basis, and we advance the costs of litigation. If we help you resolve your case, we receive a percentage of the amount you receive, and you reimburse us for the costs we advanced on your behalf.


